Supervisor of Master's Candidates
Gender:Male
Status:Employed
Department:School of Life Science and Technology
Education Level:Postgraduate (Doctoral)
Degree:Doctoral Degree in Science
Discipline:Biomedical Engineering
Paper Publications
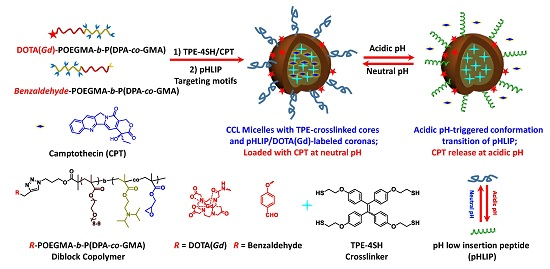
pH-Responsive Tumor-Targetable Theranostic Nanovectors Based on Core Crosslinked (CCL) Micelles with Fluorescence and Magnetic Resonance (MR) Dual Imaging Modalities and Drug Delivery Performance
Indexed by:Journal paper
Journal:polymers
Volume:8
Key Words:pH-responsive; tumor targeting; aggregation induced emission; MR imaging; pHLIP
DOI number:10.3390/polym8060226
Date of Publication:2016-06-07
Impact Factor:4.967
Abstract:The development of novel theranostic nanovectors is of particular interest in treating formidable diseases (e.g., cancers). Herein, we report a new tumor-targetable theranostic agent based on core crosslinked (CCL) micelles, possessing tumor targetable moieties and fluorescence and magnetic resonance (MR) dual imaging modalities. An azide-terminated diblock copolymer, N3-POEGMA-b-P(DPA-co-GMA), was synthesized via consecutive atom transfer radical polymerization (ATRP), where OEGMA, DPA, and GMA are oligo(ethylene glycol)methyl ether methacrylate, 2-(diisopropylamino)ethyl methacrylate, and glycidyl methacrylate, respectively. The resulting diblock copolymer was further functionalized with DOTA(Gd) (DOTA is 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetrakisacetic acid) or benzaldehyde moieties via copper(I)-catalyzed alkyne-azide cycloaddition (CuAAC) chemistry, resulting in the formation of DOTA(Gd)-POEGMA-b-P(DPA-co-GMA) and benzaldehyde-POEGMA-b-P(DPA-co-GMA) copolymers. The resultant block copolymers co-assembled into mixed micelles at neutral pH in the presence of tetrakis[4-(2-mercaptoethoxy)phenyl]ethylene (TPE-4SH), which underwent spontaneous crosslinking reactions with GMA residues embedded within the micellar cores, simultaneously switching on TPE fluorescence due to the restriction of intramolecular rotation. Moreover, camptothecin (CPT) was encapsulated into the crosslinked cores at neutral pH, and tumor-targeting pH low insertion peptide (pHLIP, sequence: AEQNPIYWARYADWLFTTPLLLLDLALLVDADEGTCG) moieties were attached to the coronas through the Schiff base chemistry, yielding a theranostic nanovector with fluorescence and MR dual imaging modalities and tumor-targeting capability. The nanovectors can be efficiently taken up by A549 cells, as monitored by TPE fluorescence. After internalization, intracellular acidic pH triggered the release of loaded CPT, killing cancer cells in a selective manner. On the other hand, the nanovectors labeled with DOTA(Gd) contrast agents exhibited increased relaxivity (r1 = 16.97 mM−1·s−1) compared to alkynyl-DOTA(Gd) small molecule precursor (r1 = 3.16 mM−1·s−1). Moreover, in vivo MRI (magnetic resonance imaging) measurements revealed CCL micelles with pHLIP peptides exhibiting better tumor accumulation and MR imaging performance as well.
Links to published journals:https://doi.org/10.3390/polym8060226
